When you think of producing a quality product what you may picture in your mind’s eye are the raw materials, equipment, methods, and all the checks that show it is indeed meeting the company standard of quality. While true, the quality of this chain of production is only as strong as the supporting sanitation links. Ideal sanitation is proactive and built into facilities from the beginning. This can mean engineering HVAC for additional air exchanges to reduce airborne particulate or designing isolated sanitizing spaces. Production systems involve many workers, materials, surfaces, high and low use equipment, exterior intake, and indoor air control. How many chances can chemical and biological contamination happen on the path to your consumer? Some examples include make-up air intakes or soil from delivery vehicle tires and footwear. These opportunities create variable types & amounts of soilage at every step of the process each representing the need for a unique sanitation standard operating procedure (SOP).
Managing sanitation requires a tailored program integrated into each level of your production. This program starts with risk-based assessments to identify the level of sanitation and special precautions needed throughout. Some areas may be low risk and suitable for every other day cleaning. Others are aseptic environments with extreme risk requiring sanitation before and after each procedure with lab testing for proper sanitation execution. Regardless of risk it is of the utmost importance to have traceability for completion of sanitation tasks to achieve a quality standard. The contribution to quality that sanitation brings is created by staff that is invested and understands its importance. For example, chocolate based cannabis edibles have many ingredients that adhere strongly to surfaces. Not only does this become an opportunity for other contamination to accumulate in this sticky matrix it also is a source of food for microbes. Sanitation of this difficult matrix requires an SOP designed for high fat and sugar content soilage executed by properly trained and equipped staff. Adherence to this SOP must be recorded by staff and consistently monitored to prevent recalls and to protect consumer health. The feedback of testing and monitoring should be quickly integrated as part of a culture of continuous process improvement.
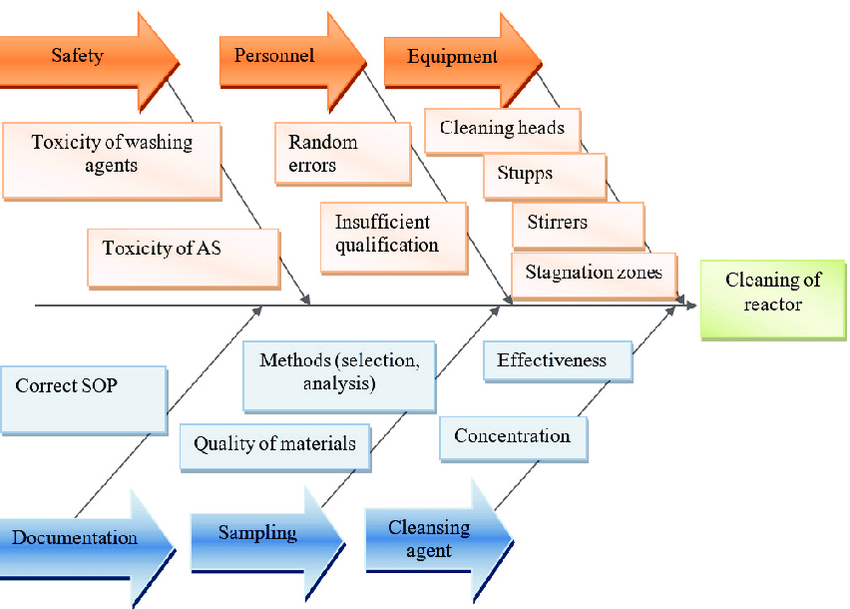
This risk-based assessment will identify specific sanitation concerns related to your product. To produce cannabis food products at scale calls for a matching sanitation regiment. It isn’t as simple as purchasing pre-mixed sanitizer every time. Doing so becomes expensive and cuts into profit margins. Best use of your chemical sanitizer and financial resources involves vetted GMP suppliers, inventory management systems, and SOP’s for each chemical sanitizing need identified during the risk assessment. Each chemical sanitizing SOP contains easily communicated instructions for dilution, use, safety, and compliance. Proper dilution is incredibly important to achieve the required action levels for sanitizing efficacy while not being over concentrated leaving a residual film. With proper dilution must come proper rinsing with monitoring. This starts with using potable water from a consistent source. The quality and properties of the water available impacts determining compatible sanitizers and should be evaluated before production. The WHO provides minimum guidelines for safe drinking water for a consistent approach across the world. Constant rinsing as part of sanitation comes with another logistical concern. Rinse water can contain strong detergents and other sanitizing agents potentially hazardous to the environment as runoff or accidental catastrophic exposure. Proper handling and disposal of cleaning waste products is an integral part of a sanitation program. Understanding the impact of run-off and the cleanings agents used is key to being good stewards of the environment and allows cannabis operators to support environmental initiatives.
There are many more aspects to sanitation to consider when achieving a quality standard, protecting consumer health, and protecting your business from audits and recalls. If you are interested in starting down the path of GMP or just looking to improve the operation of your business and need a complete sanitation program or wanting to clean yours to a higher standard contact Orion GMP solutions at [email protected]. Or visit our website and don’t forget to check out our library of resources and white papers.

Leave a Reply